Sonoporation: Underlying Mechanisms and Applications in Cellular Regulation
1First Affiliated Hospital of University of South China, Hengyang, China
2Department of Pediatrics, St Christopher’s Hospital for Children, Tower Health and Drexel University, Philadelphia, PA (S.G.)
*Correspondence: Zhiyi Chen, E-mail: zhiyi_chen@usc.edu.cn; Shuping Ge, E-mail: Shuping.Ge@towerhealth.org
Received: August 14 2020; Revised: September 5 2020; Accepted: September 10 2020; Published Online: February 6 2021
Cite this paper:
Yue Li, Zhiyi Chen and Shuping Ge. Sonoporation: Underlying Mechanisms and Applications in Cellular Regulation. BIO Integration 2021; 2(1): 29–36.
DOI: 10.15212/bioi-2020-0028. Available at: https://bio-integration.org/
Download citation
© 2021 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Ultrasound combined with microbubble-mediated sonoporation has been applied to enhance drug or gene intracellular delivery. Sonoporation leads to the formation of openings in the cell membrane, triggered by ultrasound-mediated oscillations and destruction of microbubbles. Multiple mechanisms are involved in the occurrence of sonoporation, including ultrasonic parameters, microbubbles size, and the distance of microbubbles to cells. Recent advances are beginning to extend applications through the assistance of contrast agents, which allow ultrasound to connect directly to cellular functions such as gene expression, cellular apoptosis, differentiation, and even epigenetic reprogramming. In this review, we summarize the current state of the art concerning microbubble–cell interactions and sonoporation effects leading to cellular functions.
Keywords
Microbubble, multidisciplinary, reprogramming, sonoporation, ultrasound.
Introduction
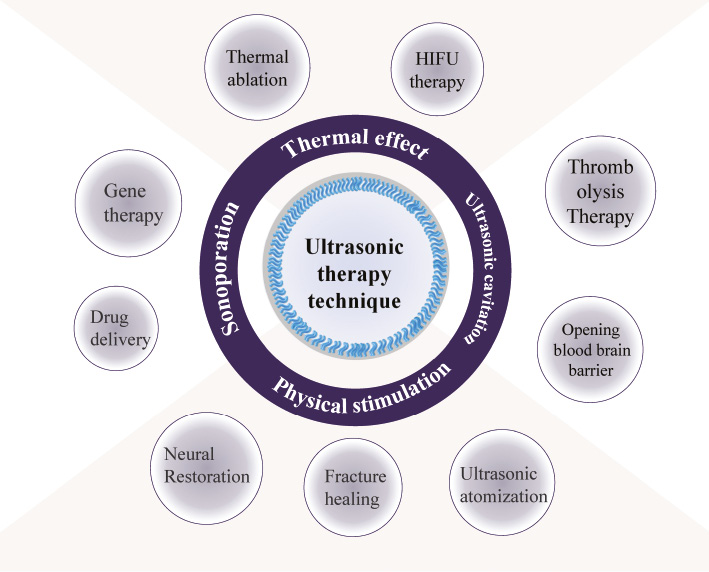
Ultrasound is widely used in clinical practice due to its advantages of being non-invasive, non-radioactive, convenient, and cost-effective [1–5]. The acoustic impedances of various tissues and organs of the human body are different, which in turn causes sound waves to generate specific echoes during the propagation in human tissues [6]. On the other hand, the safety characteristic of ultrasound application, its ability to focus on specific sites, and the potential to achieve a great diversity of therapies enrich the clinical treatment strategy [7–10] (Figure 1). For the past decades, ultrasound had shown good compatibility in combination with different technologies for disease therapy [11–15], especially ultrasound-mediated drug delivery. The key to the drug delivery system is delivering therapeutic molecules to the target area across endothelial barriers and plasma membrane to tissues or cells. Efficient aggregation at specific sites obviously reduces side effects to normal tissues. [16]. Ultrasound combined with gas-filled microbubbles is well known for its non-virus, non-invasive strategy to enhance intracellular gene or drug delivery [17–21]. By mixing cells with microbubbles, numerous temporary pores occur in the cell membrane after ultrasound exposure, which is called sonoporation [22]. The capture of the phenomenon of sonopores for the first time on the cell membrane by a scanning electron micrograph in 1999 has aroused people’s wide concern [23]. Improvement of cell membrane permeability and increase in the cellular uptake of impenetrable molecules from the extracellular matrix by sonoporation have a positive stimulation for cell growth and development [24]. Therefore, ultrasound has been utilized not only for diagnosis but also as a therapeutic modality [25].
Figure 1 Application of ultrasonic therapy technique.
However, many of the biophysical mechanisms still remain unknown. Technological innovation of ultrasound medicine has driven the development of the research in ultrasonic biophysics, which provides the necessary basic theory for the development of both diagnosis and treatment in modern ultrasound field. In this review, we state the present status of the sonoporation-mediated drug delivery system, the mechanisms of sonoporation production, and the relationship between ultrasound parameters and microbubbles. Next, we summarize the utilization of ultrasound-mediated sonoporation leading to cell endocytosis, production of reactive oxygen species (ROS), and life cycle. Finally, we discuss the role of ultrasound-mediated sonoporation in an unexplored field for cellular fate regulation.
Definition and parameters of sonoporation
Definition of Sonoporation
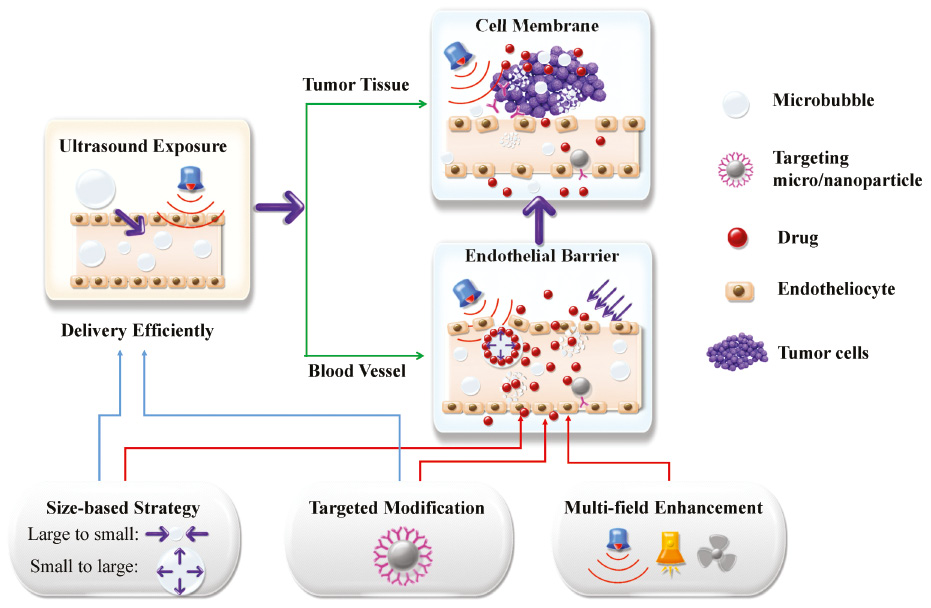
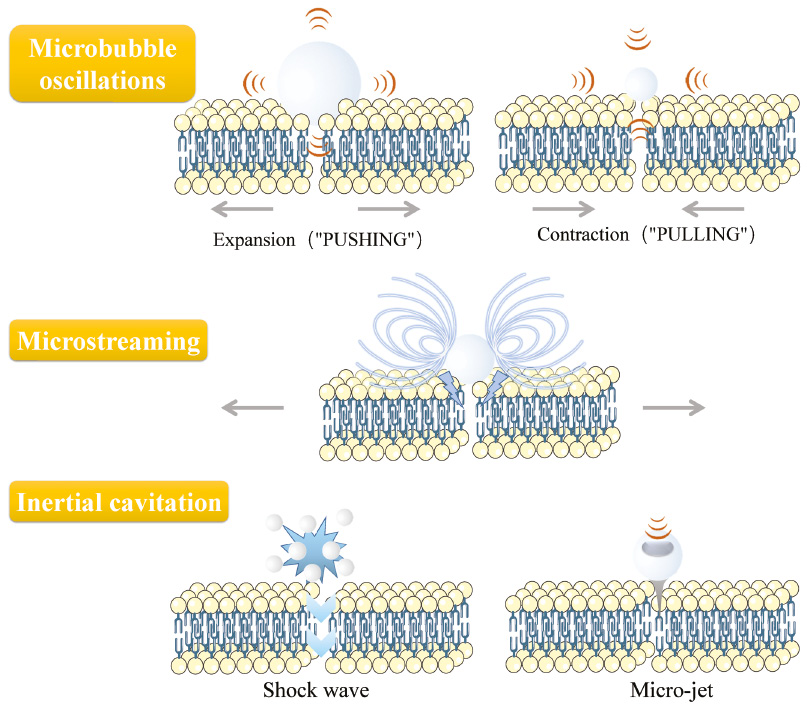
Ultrasound energy can induce temporary disruptions in the cell membrane. This manifestation of morphological changes is called sonoporation, which is generally considered as the basic principle of the ultrasound-mediated drug delivery system. The interaction of ultrasound energy with tissues or cells in vivo first induces opening endothelial junctions, and secondly reversible perforation on the cell membrane [26], stimulating cellular uptake and endocytosis [27] (Figure 2). Microbubbles usually serve as cavitation nuclei to amplify the sonoporation effects. These microbubbles are 2~8 μm in size with a biocompatible shell (lipid, protein, or polymer) [28]. The underlying mechanism of sonopore formation and cellular uptake of impermeable molecules are the activities of microbubbles on the membrane driven by ultrasound [29, 30]. Oscillating microbubbles would go through contraction and expansion and collapse with the action changing of negative acoustic pressure [31, 32]. The proximity of microbubbles to the cell surface set the surrounding liquid into motion, create shearing force to form microstreaming around the microbubbles [33, 34]. Collapsed microbubbles generate shock wave and micro-jet in the fluid (Figure 3). The deformation and collapse of microbubbles are related to the acoustic energy field, specifically to the ultrasound parameters.
Figure 2 Overview of the delivery routes enhanced by sonoporation.
Figure 3 The generation of sonoporation.
Microbubble deformation and various ultrasound parameters
Ultrasound parameters include acoustic pressure, frequency, acoustic intensity, exposure time, pulse duration, duty cycle, cavitation index (CI), mechanical index (MI), pulse repetition frequency (PRF), and others. Most studies generally indicated that acoustic pressure has a positive relationship with sonoporation-mediated delivery rate, but the opposite with cell viability [35–39]. The cavitation in fluid environment is more like a random event, which can be reduced when enough microbubbles exist simultaneously. Therefore, the concentration of microbubbles would be regarded as an optimization index for the production of sonopores and transfection efficiency [39–41]. Parameters related to cavitation, such as acoustic intensity [42–44], exposure time [39, 45], duty cycle [46], pulse duration [47], MI [48], and PRF [49] have similar trends in in vitro experiments. The opening degree of sonopores is related to the energy provided by ultrasound. The energy absorbed by the microbubbles is positively correlated with cavitation effect, which leads to more obvious sonopore formation. However, when the acoustic energy is too high, irreversible damage will be caused to the cells. This threshold depends on the tolerance and sensitivity of the cells to ultrasound. In an in vivo environment with complex conditions, the ultrasound-mediated sonoporation follows a similar rule. Shapiro et al. [39] optimized the sonoporation-based gene delivery parameters in vivo, including acoustic pressure, microbubble concentration, plasmid dosage, and irradiation time. Green fluorescent signal from the luciferase gene in the treatment group was 100-fold higher than that in the control group, which indicated that in vivo delivery efficiency is positively correlated with acoustic pressure, microbubble and plasmid DNA (pDNA) dosage, and treatment time.
Microbubble size and distance from cell membrane
The distance between the microbubbles and the cells is another consideration [50]. Qin et al. [51] revealed the impact of bubble-cell interaction parameters. The energy produced by bubble-cavitation decreases as the distance between microbubbles and cells increases. The sonoporation phenomenon cannot be detected when the microbubble-to-cell distance is larger than the microbubble’s diameter, and a larger microbubble can improve this situation, but it is easily prone to unpredictable motion when microbubbles’ diameter is larger than 5.5 μm. Furthermore, the influences of different cell lines also have been studied. Shi et al. [52] showed that four cancer cell lines (breast, ovarian, liver, and thyroid) have their own sonoporation under ultrasound treatment. The conditions (acoustic intensity, concentration of microbubbles, and treatment time) for each cancer cell line were different. This suggests that cells from different tissue sources have different sensitivity and tolerance to ultrasonic stimulation. Jia et al. [53] compared Adriamycin-resistant breast cancer MCF-7/ADR cells and MCF-7 cells at the same ultrasound exposure conditions. They found that the cell viability of both MCF-7/ADR cells and MCF-7 cells decrease with a high-intensity acoustic field, and MCF-7/ADR cells were more sensitive to ultrasound exposure.
Although the basic structure and principles of sonoporation-based delivery have been established in previous work, the efficiency of ultrasound combined with microbubbles-mediated drug or gene delivery is far from meeting clinical requirements. In-depth study of relevant underlying mechanisms and corresponding adjustments are the key to solve this limitation. Many studies in the literature show that multiple factors and mechanisms work together to explain the generation of sonoporation, including cavitation activities and biological regulatory mechanisms [54–56]. In addition, a variety of biophysical effects after sonoporation treatment, including apoptosis, actin cytoskeleton changes, and mitochondrial permeability transition pore opening, may be used as potential cellular regulatory applications [57–59].
Bioeffects of sonoporation on cells
Changes in cytoskeleton dynamics
Transformation of F-actin represents the morphological changes in the cytoskeleton, which is related to cellular motility and morphology. FITC-phalloidin can bind to F-actin to show the arrangement of the cytoskeleton. Liufu et al. found that in comparison with the control group, the sonoporation-treated group showed highlighted green fluorescent spots, which present a disorder of F-actin microfilaments [60]. The F-actin in cells aggregated and induced more dot-like structures, suggesting a complete disruption of actin microfilaments [57]. Experimental observations showed that the higher the acoustic pressure or closer the distance between the microbubble and the cell, the more obvious the pore formation occurred in the cell membrane, sometimes leading to cytoskeleton disassembly [61].
Drug delivery using sonopores or endocytosis
Sonopore formation triggers many motility mechanisms of the cellular membrane, including the movement of the cell membrane. The diameter of the sonopore has been determined (110 ± 40 nm) [62], and not all micromolecules pass through the pore pathway into the cell; endocytosis also plays an important role in ultrasound-mediated delivery. Several studies have indicated that endocytosis triggered by sonoporation may be involved in intracellular delivery of macromolecules. FITC-labeled dextran can enter the cell through sonopores (4.4 to 70 kDa) and endocytes (>155 kDa) after sonoporation. In the case of adenosine triphosphate (ATP) consumption, the delivery efficiency of the sonopore pathway decreased, and the endocytosis pathway was significantly limited, which was closely related to macropinocytosis and clathrin [63]. De et al. [64] showed that acoustic pressure is related to the approach of intracellular transport. FITC-dextran mainly enters the cell through pore formation at higher acoustic pressures, while endocytosis was more abundant at lower acoustic pressures, which was related to cytoskeletal deformation.
Production of ROS
Many studies have shown that ROS are products of the body’s normal metabolism and play an important role in human disease and aging due to their strong oxidizability. However, the imbalance between antioxidants and oxidants may induce adverse effects. The shear stress from oscillated microbubble leads to the generation of superoxide and H2O2. Jia et al. [65] assessed the degree of sonoporation and intracellular ROS changes and found that ROS production in sonopore-reversible cells increased and was positively correlated with the degree of sonoporation. Escoffre et al. also confirmed that ROS was produced during microbubble cavitation and that these reactive oxygen molecules are easily involved in cell membrane penetration and gene delivery [66].
Impact in cellular cycle function
In addition to cell membrane disruption, as a biophysical process, sonoporation induces some cellular responses, including regulation of cellular calcium ion signals [67], cell membrane potential changes [68], and cellular mechanical events at the nuclear level [69]. The cell cycle is a periodic program that leads to DNA replication and cell division [70]. As the mechanical effects of ultrasound-driven sonoporation stimulate the physical vibration of cellular structures, Fan et al. [71] investigated the influence of the cell cycle phase on the regulation of HeLa cells and their cellular responses. Their research showed that the cell cytoskeleton structural change with cell cycle phases varies, and the instantaneous biophysical effect resulted in the fastest cytoskeleton disassembly. Synchronous with the stimulation of ultrasonic microbubble-mediated sonoporation trigger, they found that disruption of the α-tubulin microtubules at the position of the microbubble located accompanied propidium iodide being delivered into the cytoplasm through the microbubble site.
Regulation of cellular fate with ultrasound
Application of cell apoptosis
After undergoing sonoporation, the appearance of cellular disruption may lead to anti-proliferation effects, for instance, causing cell apoptosis through disrupting various cell signaling pathways. Zhong et al. [72] demonstrated that sonoporation induced HL-60 cells to apoptosis possibly through the mitochondrion, where Bcl-2 decreased while Bax increased over time. The opening mitochondrial membrane through the Bax pathway released pro-apoptotic molecules (cytochrome c) into the cytoplasm, inducing cell apoptosis. However, another research [73] using sonoporation increased the percentage of apoptosis in K562 cells, and it was found that these mitochondrial membrane-depolarized cells lead to mitochondrial dysfunction, which were significantly inhibited by cyclosporine-A but not the Bax-inhibiting peptide. They proved that cytochrome c was released from the mitochondrial permeability transition pore, the proteinaceous megapore consisting of cyclophilin D, but not the Bax pathway. The specific mechanism is contradictory, and the proof of more in-depth principles remains to be revealed.
Application of stem cell-differentiation
Stem cells can differentiate into many types of cells or tissues and have an important value in clinical transplantation treatment, disease model construction, and mechanism research. In order to successfully apply stem cells to tissue engineering and regenerative medicine, the behavior of stem cells, such as responses to biochemical and biophysical cues (including adhesion, proliferation, survival, and differentiation) must be precisely regulated. Some studies have investigated if ultrasound exposure promotes neural stem cell attachment and differentiation [74]. Lee et al. showed that ultrasound significantly enhances neural stem cell and neural progenitor cell differentiation and the utilization of growth factors [75]. Cancer stem cells (CSCs), which have in common the ability to self-renew as normal stem cells, have the characteristics of stimulating tumor growth, recurrence, metastasis, and drug resistance. Differentiation of CSCs provides another therapeutic strategy that reverses the stemness of the CSC and forces it to lose its ability to self-renew [76].
Application of cellular reprogramming
In 2006, Takahashi and Yamanaka first transferred four transcription factors into mouse somatic cells through retroviral vectors and obtained induced pluripotent stem cells (iPSCs), which are very similar to embryonic stem cells (ESCs) in their morphology, as multipotent markers, and epigenetic status. The emergence of iPSCs has led to a breakthrough in the understanding of the extremely complex process of cell reprogramming [77]. The cellular signaling pathway and the pluripotency gene regulatory network together maintain the pluripotency of iPSCs. Lee et al.[78] employed biophysical stimuli such as ultrasound-mediated cellular permeation, which led to cell reprogramming, developing a non-invasive and gene/chemical-free method for multipotent cell generation from human dermal fibroblasts (HDFs). These multipotent cells exhibit a spheroid morphology, potential of differentiation, and pluripotency expression. They also found that the activation of the mitogen-activated protein kinase (MAPK) signaling pathways was closely related to cell reprogramming. Moreover, direct cell-to-cell connections facilitated reprogramming factors being released and transported into neighboring cells through ultrasound-induced sonoporation, causing a common expression of pluripotent markers and multi-lineage differentiation potential in the cell population.
Prospects and outlooks
Ultrasound is experiencing a transformation in terms of both clinical applications and technical developments. It is possible to improve the efficacy and minimize side effects by controlling sonoporation activities and designing biological regulatory strategies. Several pre-clinical studies have indicated the potential of novel ultrasound-responsive carriers to deliver multiple types of drugs including model drugs, anticancer drugs, therapeutic antibodies, genes, and nanoparticles, efficiently in various tumor models for immunotherapy [5], brain disease [79], pancreatic cancer [80], and bacterial infections [81].
Besides, one important research area encompasses the structure, morphology, and function of biological systems under ultrasound irradiation. The inherent advantage of ultrasound is that it applies basic physical stimulation to living tissue. The fields of molecular ultrasound and ultrasound genetics seek to link these physical forces with the functions of biomolecules and cells to achieve precise control of cells [82, 83]. In addition, genetic engineering editing allows biomolecules to interact with ultrasound. Ultrasound presents as both a waveform and a sound energy form. For example, the thermal effect or mechanical effect of high-intensity focused ultrasound (HIFU) can be used to locally elevate tissue temperature and activate temperature-sensitive proteins and pathways [84, 85]. Although this field is still in its infancy, it has opened the door to precise ultrasound control. As an acoustic waveform, it can detect loading signals; as an energy form, it can change the medium and microenvironment for cell survival [86–88]. Although cavitation-induced sonoporation has the potential for drug and gene delivery, the cavitation events seem to be a random process, causing a low consistent sonoporation outcome. The elucidation of all of the interactions between microbubbles and cells would help to improve the uniformity, efficiency, and safety of the sonoporation. Multidisciplinary technology integration should be advocated and promoted to address this limitation, including the machining device [89], biophysics [56], physics [90], and sonochemistry [91]. We believe that ultrasound will have more extensive research in the future, providing more options for targeted regulation and treatment protocol as well as revealing more biophysical mechanisms.
References
- Carpentier A, Canney M, Vignot A, Reina V, Beccaria K, et al. Clinical trial of blood-brain barrier disruption by pulsed ultrasound. Sci Transl Med 2016;8:343re2. [PMID: 27306666 DOI: 10.1126/scitranslmed.aaf6086]
- Dimcevski G, Kotopoulis S, Bjanes T, Hoem D, Schjott J, et al. A human clinical trial using ultrasound and microbubbles to enhance gemcitabine treatment of inoperable pancreatic cancer. J Control.Release 2016;243:172-81. [PMID: 27744037 DOI: 10.1016/j.jconrel.2016.10.007]
- Tang W, Yang Z, Wang S, Wang Z, Song J, et al. Organic semiconducting photoacoustic nanodroplets for laser-activatable ultrasound imaging and combinational cancer therapy. ACS Nano 2018;12:2610-22. [PMID: 29451774 DOI: 10.1021/acsnano.7b08628]
- Willmann JK, Bonomo L, Testa AC, Rinaldi P, Rindi G, et al. Ultrasound molecular imaging with BR55 in patients with breast and ovarian lesions: first-in-human results. J Clin Oncol 2017;35:2133-40. [PMID: 28291391 DOI: 10.1200/JCO.2016.70.8594]
- Yang C, Li Y, Du M, Chen Z. Recent advances in ultrasound-triggered therapy. J Drug Target 2019;27:33-50. [PMID: 29659307 DOI: 10.1080/1061186X.2018.1464012]
- Jaeger M, Held G, Peeters S, Preisser S, Gruenig M, et al. Computed ultrasound tomography in echo mode for imaging speed of sound using pulse-echo sonography: proof of principle. Ultrasound Med Biol 2015;41:235-50. [PMID: 25220274 DOI: 10.1016/j.ultrasmedbio.2014.05.019]
- Canavese G, Ancona A, Racca L, Canta M, Dumontel B, et al. Nanoparticle-assisted ultrasound: a special focus on sonodynamic therapy against cancer. Chem Eng Trans 2018;340:155-72. [PMID: 30881202 DOI: 10.1016/j.cej.2018.01.060]
- Sennoga CA, Kanbar E, Auboire L, Dujardin P-A, Fouan D, et al. Microbubble-mediated ultrasound drug-delivery and therapeutic monitoring. Expert Opin Drug Deliv 2017;14:1031-43. [PMID: 27892760 DOI: 10.1080/17425247.2017.1266328]
- Wu S-Y, Aurup C, Sanchez CS, Grondin J, Zheng W, et al. Efficient blood-brain barrier opening in primates with neuronavigation-guided ultrasound and real-time acoustic mapping. Sci Rep 2018;8:7978. [PMID: 29789530 DOI: 10.1038/s41598-018-25904-9]
- Zhang L, Yin T, Li B, Zheng R, Qiu C, et al. Size-modulable nanoprobe for high-performance ultrasound imaging and drug delivery against cancer. ACS Nano 2018;12:3449-60. [PMID: 29634240 DOI: 10.1021/acsnano.8b00076]
- Bez M, Foiret J, Shapiro G, Pelled G, Ferrara KW, et al. Nonviral ultrasound-mediated gene delivery in small and large animal models. Nat Protoc 2019;14:1015-26. [PMID: 30804568 DOI: 10.1038/s41596-019-0125-y]
- Negishi Y, Endo-Takahashi Y, Maruyama K. Gene delivery systems by the combination of lipid bubbles and ultrasound. Drug Discov Ther 2016;10:248-55. [PMID: 27795481 DOI: 10.5582/ddt.2016.01063]
- Suzuki R, Oda Y, Utoguchi N, Maruyama K. Progress in the development of ultrasound-mediated gene delivery systems utilizing nano- and microbubbles. J Control Release 2011;149:36-41. [PMID: 20470839 DOI: 10.1016/j.jconrel.2010.05.009]
- Xia H, Zhao Y, Tong R. Ultrasound-mediated polymeric micelle drug delivery. In: Escoffre JM, Bouakaz A, editors. Therapeutic ultrasound. Advances in Experimental Medicine and Biology. 2016;880:365-84. [PMID: 26486348 DOI: 10.1007/978-3-319-22536-4_20]
- Yoon CS, Park JH. Ultrasound-mediated gene delivery. Expert Opin Drug Deliv 2010;7:321-30. [PMID: 20166854 DOI: 10.1517/17425241003596329]
- Zhang Y, Chan HF, Leong KW. Advanced materials and processing for drug delivery: the past and the future. Adv Drug Deliv Rev 2013;65:104-20. [PMID: 23088863 DOI: 10.1016/j.addr.2012.10.003]
- Klibanov AL. Microbubble contrast agents – targeted ultrasound imaging and ultrasound-assisted drug-delivery applications. Invest Radiol 2006;41:354-62. [PMID: 16481920 DOI: 10.1097/01.rli.0000199292.88189.0f]
- Ferrara K, Pollard R, Borden M. Ultrasound microbubble contrast agents: fundamentals and application to gene and drug delivery. Annu Rev Biomed Eng 2007;9:415-47. [PMID: 17651012 DOI: 10.1146/annurev.bioeng.8.061505.095852]
- Newman CMH, Bettinger T. Gene therapy progress and prospects: ultrasound for gene transfer. Gene Ther 2007;14:465-75. [PMID: 17339881 DOI: 10.1038/sj.gt.3302925]
- Luo WX, Wen G, Yang L, Tang J, Wang JG, et al. Dual-targeted and pH-sensitive doxorubicin prodrug-microbubble complex with ultrasound for tumor treatment. Theranostics 2017;7:452-65. [PMID: 28255342 DOI: 10.7150/thno.16677]
- Jones RM, Deng LL, Leung K, McMahon D, O’Reilly MA, et al. Three-dimensional transcranial microbubble imaging for guiding volumetric ultrasound-mediated blood-brain barrier opening. Theranostics 2018;8:2909-26. [PMID: 29896293 DOI: 10.7150/thno.24911]
- Bao SP, Thrall BD, Miller DL. Transfection of a reporter plasmid into cultured cells by sonoporation in vitro. Ultrasound Med Biol 1997;23:953-9. [PMID: 9300999 DOI: 10.1016/s0301-5629(97)00025-2]
- Tachibana K, Uchida T, Ogawa K, Yamashita N, Tamura K. Induction of cell-membrane porosity by ultrasound. Lancet 1999;353:1409. [PMID: 10227224 DOI: 10.1016/S0140-6736(99)01244-1]
- Lin YT, Lin LZ, Cheng MW, Jin LF, Du LF, et al. Effect of acoustic parameters on the cavitation behavior of SonoVue microbubbles induced by pulsed ultrasound. Ultrason Sonochem 2017;35:176-84. [PMID: 27707644 DOI: 10.1016/j.ultsonch.2016.09.016]
- Schandelmaier S, Kaushal A, Lytvyn L, Heels-Ansdell D, Siemieniuk RAC, et al. Low intensity pulsed ultrasound for bone healing: systematic review of randomized controlled trials. Br Med J 2017;356. [PMID: 28348110 DOI: 10.1136/bmj.j656]
- van Rooij T, Skachkov I, Beekers I, Lattwein KR, Voorneveld JD, et al. Viability of endothelial cells after ultrasound-mediated sonoporation: influence of targeting, oscillation, and displacement of microbubbles. J Control Release 2016;238:197-211. [PMID: 27469471 DOI: 10.1016/j.jconrel.2016.07.037]
- Delalande A, Leduc C, Midoux P, Postema M, Pichon C. Efficient gene delivery by sonoporation is associated with microbubble entry into cells and the clathrin-dependent endocytosis pathway. Ultrasound Med Biol 2015;41:1913-26. [PMID: 25929996 DOI: 10.1016/j.ultrasmedbio.2015.03.010]
- Yue L, Chen Y, Meng D, Chen ZY. Ultrasound technology for molecular imaging: from contrast agents to multimodal imaging. ACS Biomater Sci Eng 2018;4:2716-28:acsbiomaterials.8b00421. [DOI: 10.1021/acsbiomaterials.8b00421]
- Kooiman K, Roovers S, Langeveld SAG, Kleven RT, Dewitte H, et al. Ultrasound-responsive cavitation nuclei for therapy and drug delivery. Ultrasound Med Biol 2020;46:1296-325. [PMID: 32165014 DOI: 10.1016/j.ultrasmedbio.2020.01.002]
- Lentacker I, De Cock I, Deckers R, De Smedt SC, Moonen CTW. Understanding ultrasound induced sonoporation: definitions and underlying mechanisms. Adv Drug Deliv Rev 2014;72:49-64. [PMID: 24270006 DOI: 10.1016/j.addr.2013.11.008]
- Sboros V. Response of contrast agents to ultrasound. Adv Drug Deliv Rev 2008;60:1117-36. [[PMID: 18486270 DOI: 10.1016/j.addr.2008.03.011]
- Delalande A, Kotopoulis S, Postema M, Midoux P, Pichon C. Sonoporation: mechanistic insights and ongoing challenges for gene transfer. Gene 2013;525:191-9. [PMID: 23566843 DOI: 10.1016/j.gene.2013.03.095]
- Cleve S, Guedra M, Mauger C, Inserra C, Blanc-Benon P. Microstreaming induced by acoustically trapped, non-spherically oscillating microbubbles. J Fluid Mech 2019;875:597-621. [DOI: 10.1017/jfm.2019.511]
- Collis J, Manasseh R, Liovic P, Tho P, Ooi A, et al. Cavitation microstreaming and stress fields created by microbubbles. Ultrasonics 2010;50:273-9. [PMID: 19896683 DOI: 10.1016/j.ultras.2009.10.002]
- Escoffre J-M, Novell A, Piron J, Zeghimi A, Doinikov A, et al. Microbubble attenuation and destruction: are they involved in sonoporation efficiency? IEEE Trans Ultrason Ferroelectr Freq Control 2013;60:46-52. [PMID: 23287912 DOI: 10.1109/TUFFC.2013.2536]
- Lamanauskas N, Novell A, Escoffre JM, Venslauskas M, Satkauskas S, et al. Bleomycin delivery into cancer cells in vitro with ultrasound and SonoVue (R) or BR14 (R) microbubbles. J Drug Target 2013;21:407-14. [PMID: 23336182 DOI: 10.3109/1061186X.2012.761223]
- Qiu Y, Zhang C, Tu J, Zhang D. Microbubble-induced sonoporation involved in ultrasound-mediated DNA transfection in vitro at low acoustic pressures. J Biomech 2012;45:1339-45. [PMID: 22498312 DOI: 10.1016/j.jbiomech.2012.03.011]
- Rong N, Zhou H, Liu R, Wang Y, Fan Z. Ultrasound and microbubble mediated plasmid DNA uptake: a fast, global and multi-mechanisms involved process. J Control Release 2018;273:40-50. [PMID: 29407677 DOI: 10.1016/j.jconrel.2018.01.014]
- Shapiro G, Wong AW, Bez M, Yang F, Tam S, et al. Multiparameter evaluation of in vivo gene delivery using ultrasound-guided, microbubble-enhanced sonoporation. J Control Release 2016;223:157-64. [PMID: 26682505 DOI: 10.1016/j.jconrel.2015.12.001]
- Han YW, Ikegami A, Chung P, Zhang L, Deng CX. Sonoporation is an efficient tool for intracellular fluorescent dextran delivery and one-step double-crossover mutant construction in Fusobactetium nucleatum. Appl Environ Microbiol 2007;73:3677-83. [PMID: 17449701 DOI: 10.1128/AEM.00428-07]
- Qu N, Shi DD, Shang MM, Duan SJ, Guo L, et al. Breast cancer cell line phenotype affects sonoporation efficiency under optimal ultrasound microbubble conditions. Med Sci Monit 2018;24:9054-62. [PMID: 30546004 DOI: 10.12659/MSM.910790]
- Lo C-W, Desjouy C, Chen S-R, Lee J-L, Inserra C, et al. Stabilizing in vitro ultrasound-mediated gene transfection by regulating cavitation. Ultrason Sonochem 2014;21:833-9. [PMID: 24216067 DOI: 10.1016/j.ultsonch.2013.10.017]
- Jia YL, Yuan WJ, Zhang K, Wang J, Wang P, et al. Comparison of cell membrane damage induced by the therapeutic ultrasound on human breast cancer MCF-7 and MCF-7/ADR cells. Ultrason Sonochem 2015;26:128-35. [PMID: 25771334 DOI: 10.1016/j.ultsonch.2015.03.001]
- Yamashita T, Ando K. Low-intensity ultrasound induced cavitation and streaming in oxygen-supersaturated water: role of cavitation bubbles as physical cleaning agents. Ultrason Sonochem 2019;52:268-79. [PMID: 30573434 DOI: 10.1016/j.ultsonch.2018.11.025]
- Liao ZK, Tsai KC, Wang HT, Tseng SH, Deng WP, et al. Sonoporation-mediated anti-angiogenic gene transfer into muscle effectively regresses distant orthotopic tumors. Cancer Gene Ther 2012;19:171-80. [[PMID: 22095386 DOI: 10.1038/cgt.2011.73]
- Bazan-Peregrino M, Arvanitis CD, Rifai B, Seymour LW, Coussios C-C. Ultrasound-induced cavitation enhances the delivery and therapeutic efficacy of an oncolytic virus in an in vitro model. J Control Release 2012;157:235-42. [PMID: 21982902 DOI: 10.1016/j.jconrel.2011.09.086]
- Tran DM, Harrang J, Song S, Chen J, Smith BM, et al. Prolonging pulse duration in ultrasound-mediated gene delivery lowers acoustic pressure threshold for efficient gene transfer to cells and small animals. J Control Release 2018;279:345-54. [PMID: 29702143 DOI: 10.1016/j.jconrel.2018.04.012]
- Afadzi M, Strand SP, Nilssen EA, Masoy S-E, Johansen TF, et al. Mechanisms of the ultrasound-mediated intracellular delivery of liposomes and dextrans. IEEE Trans Ultrason Ferroelectr Freq Control 2013;60:21-33. [PMID: 23287910 DOI: 10.1109/TUFFC.2013.2534]
- Cheng M, Li F, Han T, Yu ACH, Qin P. Effects of ultrasound pulse parameters on cavitation properties of flowing microbubbles under physiologically relevant conditions. Ultrason Sonochem 2019;52:512-21. [PMID: 30642801 DOI: 10.1016/j.ultsonch.2018.12.031]
- Zhou Y, Yang K, Cui J, Ye JY, Deng CX. Controlled permeation of cell membrane by single bubble acoustic cavitation. J Control Release 2012;157:103-11. [PMID: 21945682 DOI: 10.1016/j.jconrel.2011.09.068]
- Qin P, Xu L, Han T, Du L, Yu ACH. Effect of non-acoustic parameters on heterogeneous sonoporation mediated by single-pulse ultrasound and microbubbles. Ultrason Sonochem 2016;31:107-15. [PMID: 26964929 DOI: 10.1016/j.ultsonch.2015.12.001]
- Shi D, Guo L, Duan S, Shang M, Meng D, et al. Influence of tumor cell lines derived from different tissue on sonoporation efficiency under ultrasound microbubble treatment. Ultrason Sonochem 2017;38:598-603. [PMID: 27562907 DOI: 10.1016/j.ultsonch.2016.08.022]
- Jia Y, Yuan W, Zhang K, Wang J, Wang P, et al. Comparison of cell membrane damage induced by the therapeutic ultrasound on human breast cancer MCF-7 and MCF-7/ADR cells. Ultrason Sonochem 2015;26:128-35. [PMID: 25771334 DOI: 10.1016/j.ultsonch.2015.03.001]
- Bouakaz A, Zeghimi A, Doinikov AA. Sonoporation: concept and mechanisms. In: Escoffre JM, Bouakaz A, editors. Therapeutic ultrasound. Advances in Experimental Medicine and Biology. 2016;880:175-89.
- Guo X, Cai C, Xu G, Yang Y, Tu J, et al. Interaction between cavitation microbubble and cell: a simulation of sonoporation using boundary element method (BEM). Ultrason Sonochem 2017;39:863-71. [PMID: 28733016 DOI: 10.1016/j.ultsonch.2017.06.016]
- Helfield B, Chen X, Watkins SC, Villanueva FS. Biophysical insight into mechanisms of sonoporation. Proc Natl Acad Sci USA 2016;113:9983-8. [PMID: 27551081 DOI: 10.1073/pnas.1606915113]
- Li Y, Wang P, Chen X, Hu J, Liu Y, et al. Activation of microbubbles by low-intensity pulsed ultrasound enhances the cytotoxicity of curcumin involving apoptosis induction and cell motility inhibition in human breast cancer MDA-MB-231 cells. Ultrason Sonochem 2016;33:26-36. [PMID: 27245953 DOI: 10.1016/j.ultsonch.2016.04.012]
- Qin P, Han T, Yu ACH, Xu L. Mechanistic understanding the bioeffects of ultrasound-driven microbubbles to enhance macromolecule delivery. J Control Release 2018;272:169-81. [PMID: 29305924 DOI: 10.1016/j.jconrel.2018.01.001]
- Yu H, Xu L. Cell experimental studies on sonoporation: state of the art and remaining problems. J Control Release 2014;174:151-60. [PMID: 24291334 DOI: 10.1016/j.jconrel.2013.11.010]
- Liufu C, Li Y, Lin Y, Yu J, Du M, et al. Synergistic ultrasonic biophysical effect-responsive nanoparticles for enhanced gene delivery to ovarian cancer stem cells. Drug Deliv 2020;27:1018-33. [PMID: 32627597 DOI: 10.1080/10717544.2020.1785583]
- Wang M, Zhang Y, Cai C, Tu J, Guo X, et al. Sonoporation-induced cell membrane permeabilization and cytoskeleton disassembly at varied acoustic and microbubble-cell parameters. Sci Rep 2018;8:3885. [PMID: 29497082 DOI: 10.1038/s41598-018-22056-8]
- Zhou Y, Kumon RE, Cui J, Deng CX. The size of sonoporation pores on the cell membrane. Ultrasound Med Biol 2009;35:1756-60. [PMID: 19647924 DOI: 10.1016/j.ultrasmedbio.2009.05.012]
- Meijering BDM, Juffermans LJM, van Wamel A, Henning RH, Zuhorn IS, et al. Ultrasound and microbubble-targeted delivery of macromolecules is regulated by induction of endocytosis and pore formation. Circ Res 2009;104:679-87. [PMID: 19168443 DOI: 10.1161/CIRCRESAHA.108.183806]
- De Cock I, Zagato E, Braeckmans K, Luan Y, de Jong N, et al. Ultrasound and microbubble mediated drug delivery: acoustic pressure as determinant for uptake via membrane pores or endocytosis. J Control Release 2015;197:20-8. [PMID: 25449801 DOI: 10.1016/j.jconrel.2014.10.031]
- Jia C, Xu L, Han T, Cai P, Yu ACH, et al. Generation of reactive oxygen species in heterogeneously sonoporated cells by microbubbles with single-pulse ultrasound. Ultrasound Med Biol 2018;44:1074-85. [PMID: 29499918 DOI: 10.1016/j.ultrasmedbio.2018.01.006]
- Escoffre J-M, Campomanes P, Tarek M, Bouakaz A. New insights on the role of ROS in the mechanisms of sonoporation-mediated gene delivery. Ultrason Sonochem 2020;64:104998. [PMID: 32062534 DOI: 10.1016/j.ultsonch.2020.104998]
- Park J, Fan Z, Kumon RE, El-Sayed MEH, Deng CX. Modulation of intracellular Ca2+ concentration in brain microvascular endothelial cells in vitro by acoustic cavitation. Ultrasound Med Biol 2010;36:1176-87. [PMID: 20620704 DOI: 10.1016/j.ultrasmedbio.2010.04.006]
- Tran TA, Le Guennec JY, Bougnoux P, Tranquart F, Bouakaz A. Characterization of cell membrane response to ultrasound activated microbubbles. IEEE Trans Ultrason Ferroelectr Freq Control 2008;55:43-9. [PMID: 18334312 DOI: 10.1109/TUFFC.2008.615]
- Chen X, Leow RS, Hu Y, Wan JMF, Yu ACH. Single-site sonoporation disrupts actin cytoskeleton organization. J R Soc Interface 2014;11:20140071. [PMID: 24671936 DOI: 10.1098/rsif.2014.0071]
- Jones MC, Zha J, Humphries MJ. Connections between the cell cycle, cell adhesion and the cytoskeleton. Philos Trans R Soc Lond, B, Biol Sci 2019;374:20180227. [PMID: 31431178 DOI: 10.1098/rstb.2018.0227]
- Fan P, Zhang Y, Guo X, Cai C, Wang M, et al. Cell-cycle-specific cellular responses to sonoporation. Theranostics 2017;7:4894-908. [PMID: 29187912 DOI: 10.7150/thno.20820]
- Zhong W, Sit WH, Wan JMF, Yu ACH. Sonoporation induces apoptosis and cell cycle arrest in human promyelocytic leukemia cells. Ultrasound Med Biol 2011;37:2149-59. [PMID: 22033133 DOI: 10.1016/j.ultrasmedbio.2011.09.012]
- Zhao L, Feng Y, Shi A, Zong Y, Wan M. Apoptosis induced by microbubble-assisted acoustic cavitation in K562 cells: the predominant role of the cyclosporin A-dependent mitochondrial permeability transition pore. Ultrasound Med Biol 2015;41:2755-64. [PMID: 26164288 DOI: 10.1016/j.ultrasmedbio.2015.05.021]
- Lee IC, Lo T-L, Young T-H, Li Y-C, Chen NG, et al. Differentiation of neural stem/progenitor cells using low-intensity ultrasound. Ultrasound Med Biol 2014;40:2195-206. [PMID: 25023110 DOI: 10.1016/j.ultrasmedbio.2014.05.001]
- Lee IC, Wu H-J, Liu H-L. Dual-frequency ultrasound induces neural stem/progenitor cell differentiation and growth factor utilization by enhancing stable cavitation. ACS Chem Neurosci 2019;10:1452-61. [PMID: 30608667 DOI: 10.1021/acschemneuro.8b00483]
- Lee IC, Fadera S, Liu H-L. Strategy of differentiation therapy: effect of dual-frequency ultrasound on the induction of liver cancer stem-like cells on a HA-based multilayer film system. J Mater Chem B 2019;7:5401-11. [PMID: 31414097 DOI: 10.1039/c9tb01120j]
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006;126:663-76. [PMID: 16904174 DOI: 10.1016/j.cell.2006.07.024]
- Lee YS, Heo H, Lee J, Moon SU, Jung WY, et al. An ultra-effective method of generating extramultipotent cells from human fibroblasts by ultrasound. Biomaterials 2017;143:65-78. [PMID: 28763631 DOI: 10.1016/j.biomaterials.2017.07.033]
- Mead BP, Kim N, Miller GW, Hodges D, Mastorakos P, et al. Novel focused ultrasound gene therapy approach noninvasively restores dopaminergic neuron function in a rat Parkinson’s disease model. Nano Lett 2017;17:3533-42. [PMID: 28511006 DOI: 10.1021/acs.nanolett.7b00616]
- Nesbitt H, Sheng Y, Kamila S, Logan K, Thomas K, et al. Gemcitabine loaded microbubbles for targeted chemo-sonodynamic therapy of pancreatic cancer. J Control Release 2018;279:8-16.[PMID: 29653222 DOI: 10.1016/j.jconrel.2018.04.018]
- Horsley H, Owen J, Browning R, Carugo D, Malone-Lee J, et al. Ultrasound-activated microbubbles as a novel intracellular drug delivery system for urinary tract infection. J Control Release 2019;301:166-75. [PMID: 30904501 DOI: 10.1016/j.jconrel.2019.03.017]
- Huang P. An integrated approach to ultrasound imaging in medicine and biology. BIO Integration 2020;1. [DOI: 10.15212/bioi-2020-0036]
- Hansen-Bruhn M, de Ávila BE-F, Beltrán-Gastélum M, Zhao J, Ramírez-Herrera DE, et al. Active intracellular delivery of a Cas9/sgRNA complex using ultrasound-propelled nanomotors. Angew Chem Int Ed Engl 2018;57:2657-61. [PMID: 29325201 DOI: 10.1002/anie.201713082]
- Bourdeau RW, Lee-Gosselin A, Lakshmanan A, Farhadi A, Kumar SR, et al. Acoustic reporter genes for noninvasive imaging of microorganisms in mammalian hosts. Nature 2018;553:86-90. [PMID: 29300010 DOI: 10.1038/nature25021]
- Lipsman N, Meng Y, Bethune AJ, Huang Y, Lam B, et al. Blood-brain barrier opening in Alzheimer’s disease using MR-guided focused ultrasound. Nat Commun 2018;9:2336. [PMID: 30046032 DOI: 10.1038/s41467-018-04529-6]
- Kooiman K, Vos HJ, Versluis M, de Jong N. Acoustic behavior of microbubbles and implications for drug delivery. Adv Drug Deliv Rev 2014;72:28-48. [PMID: 24667643 DOI: 10.1016/j.addr.2014.03.003]
- Leinenga G, Langton C, Nisbet R, Gotz J. Ultrasound treatment of neurological diseases – current and emerging applications. Nat Rev Neurol 2016;12:161-74. [PMID: 26891768 DOI: 10.1038/nrneurol.2016.13]
- Boissenot T, Bordat A, Fattal E, Tsapis N. Ultrasound-triggered drug delivery for cancer treatment using drug delivery systems: from theoretical considerations to practical applications. J Control Release 2016;241:144-63. [PMID: 27667179 DOI: 10.1016/j.jconrel.2016.09.026]
- Meng L, Liu X, Wang Y, Zhang W, Zhou W, et al. Sonoporation of cells by a parallel stable cavitation microbubble array. Adv Sci (Weinh) 2019;6:1900557. [PMID: 31508275 DOI: 10.1002/advs.201900557]
- Helfield BL, Chen X, Qin B, Watkins SC, Villanueva FS. Mechanistic insight into sonoporation with ultrasound-stimulated polymer microbubbles. Ultrasound Med Biol 2017;43:2678-89. [PMID: 28847500 DOI: 10.1016/j.ultrasmedbio.2017.07.017]
- Keller S, Bruce M, Averkiou MA. Ultrasound imaging of microbubble activity during sonoporation pulse sequences. Ultrasound Med Biol 2019;45:833-45. [PMID: 30638695 DOI: 10.1016/j.ultrasmedbio.2018.11.011]